Federal Appeals Court Reinstates In-Person Requirement for Abortion Pill, Limiting Mail-Order Access
May 1, 2026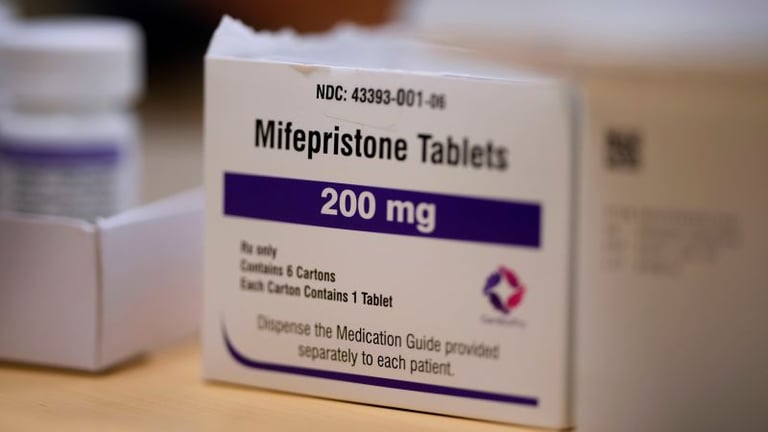
A federal appeals court in the Fifth Circuit has reinstated the in-person dispensing requirement for mifepristone, reversing a broader move toward mail-order access and potentially limiting access to medication abortion.
The panel’s decision, written by Circuit Judge Kyle Duncan and joined by two others, arises from Louisiana’s lawsuit challenging the FDA’s 2023 rule that allowed mail-order distribution of mifepristone and telehealth prescribing.
With the stay in place on appeal, the ruling could block mailed prescriptions unless stayed by higher courts, prompting potential emergency action at the Supreme Court.
This story is part of a broader news briefing that covers health and policy topics in the program, signaling ongoing coverage of abortion access and regulatory issues.
Related program segments indicate the broadcast is including broader health, policy, and lifestyle coverage alongside this case.
Judges noted uncertainty about when the FDA would complete its safety review of mifepristone and emphasized ongoing data collection as part of the ruling.
Advocacy groups reacted variably: reproductive-rights supporters warned the ruling would reduce access for rural, low-income, and marginalized communities, while abortion opponents praised stricter safety oversight.
Mifepristone is presented as a medically safe option with comparatively few side effects, underscoring public health considerations in the regulatory dispute.
This case is part of a broader line of lawsuits challenging the drug’s initial approval and subsequent rules that expanded access.
The ruling questions the FDA’s authority to overrule state bans and notes the agency cannot guarantee a timely safety review, suggesting possible broader impact beyond states with explicit bans, including telemedicine access in permissive states.
The dispute highlights ongoing tensions among abortion access, state restrictions, and federal regulation in the wake of the Dobbs decision.
Other anti-abortion states have pursued similar challenges to FDA regulations, while earlier court actions paused the mail-order rule during the COVID-era safety reviews.
Summary based on 7 sources
Get a daily email with more US News stories
Sources

NBC News • May 1, 2026
Appeals court reinstates requirement that abortion pills be dispensed in person
Los Angeles Times • May 1, 2026
Court restricts abortion access across U.S. by blocking the mailing of mifepristone - Los Angeles Times
