New Drug GLP-1–GIP–Lani Shows Promising Results for Obesity and Diabetes Treatment in Mice
April 29, 2026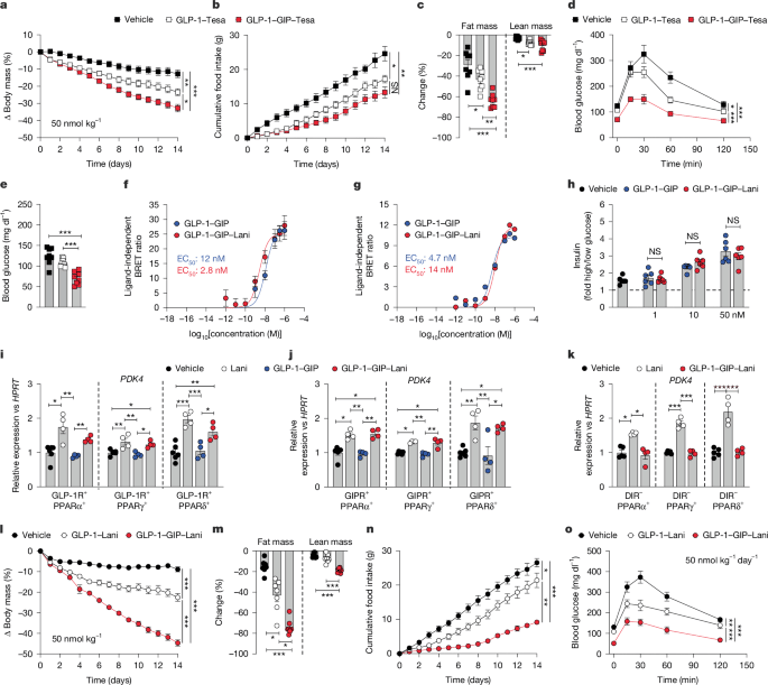
A unimolecular quintuple agonist, GLP-1R–GIPR co-agonism combined with PPARα/β/δ agonism (via Lani) delivered to GLP-1R/GIPR-expressing cells shows enhanced metabolic efficacy in obese and insulin-resistant mice versus GLP-1R–GIPR co-agonism and semaglutide.
Overall, GLP-1–GIP–Lani presents substantial therapeutic value for obesity and diabetes by combining targeted incretin signaling with PPAR pathways.
In vivo, GLP-1–GIP–Lani achieves greater reductions in body weight, food intake, and hyperglycemia than GLP-1R–GIPR co-agonism and semaglutide, with effects attenuated in mice lacking GLP-1R, GIPR, or PPARδ, and absent in DIR-KO mice, indicating dependency on these targets.
Safety assessments show no renal toxicity and no anemia or edema; cardiovascular metrics improve with preserved blood pressure, and weight loss effects persist in obesity-prone db/db mice.
Adipose tissue analysis indicates GLP-1–GIP–Lani does not drive adipocyte differentiation but enhances glucose uptake into BAT and other tissues, without upregulating thermogenic genes in BAT.
GLP-1–GIP–Lani improves glucose tolerance and lowers hepatic gluconeogenesis (Pcx, Pepck1) with anti-inflammatory transcriptomic effects in liver and skeletal muscle.
Compared with weight-matched GLP-1–GIP-treated controls, GLP-1–GIP–Lani provides superior glycemic benefits beyond weight loss, suggesting weight-independent glucose-lowering mechanisms.
Dose-ranging identifies 10 nmol/kg as a lead dose in diet-induced obesity mice, with GLP-1–GIP–Lani outperforming semaglutide on weight, fat mass, glucose metrics, and insulin sensitivity.
Transgenic adipose-GIPR overexpression enhances weight loss with incretin therapies; GLP-1–GIP–Lani shows robust, though not uniquely superior, effects in GIPR-overexpressing models.
Mechanistic knockout data show GLP-1–GIP–Lani’s glucose-lowering effect depends on PPARδ and incretin receptor signaling, with complete dependence in double-receptor knockout models.
In vitro, GLP-1–GIP–Lani signaling through incretin receptors mirrors GLP-1R–GIPR co-agonism, both driving glucose-stimulated insulin secretion; GLP-1–GIP–Lani also activates PPAR target genes in a receptor-dependent manner.
Summary based on 1 source
Get a daily email with more Science stories
Source
Nature • Apr 29, 2026
GLP-1R–GIPR–PPARα/γ/δ quintuple agonism corrects obesity and diabetes in mice