New Study Maps Kidney Niches in Diabetic Kidney Disease, Unveils Targets for Therapies
April 29, 2026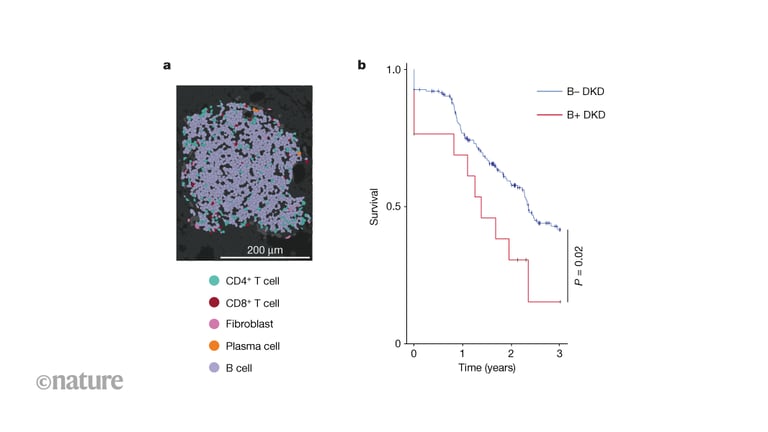
In DKD, integrating spatial transcriptomics with single-cell references defines kidney niches and microenvironments, revealing profibrotic remodeling and a B cell–rich subset with potential for targeted therapy and biomarker development.
The study identifies 20 cell types across glomerular, vascular, tubular, and immune domains, enabling refined annotation of structures such as glomeruli, LoH segments, and injured tubule states with high-resolution data.
Spatial mapping links DKD severity to immune–tubular interactions, showing reduced inflammation-resolving ANXA1–FPR3 signaling and increased CXCL12–CXCR4 and TNFSF13–TNFRSF13B interactions that recruit plasma cells, reflecting dynamic immune modulation through disease stages.
Kidney niches were defined by aggregating cell–cell relationships within 20–80 micrometers, yielding 11 recurring niches (including glomerular, vascular, PT/DCT/CNT, and injury-associated iPT/iTAL) that mirror kidney architecture while highlighting disease-related organization.
Profibrotic microenvironments emerge in injured tubules in DKD, with fibroblasts and immune cells driving extracellular matrix remodeling, inflammation, and neovascularization; ligand–receptor analyses implicate HAVCR1, EGFR signaling, JAG2–NOTCH, and PDGF pathways in fibrotic progression.
A spatial single-cell atlas of human DKD was created from 64 FFPE kidney samples across 58 patients, profiling over 5 million cells with CosMx and Xenium and integrating with a reference snRNA-seq atlas.
Bulk RNA-seq validation across 843 kidney samples supports niche and interaction findings, with correlations between specific ligand–receptor pairs and disease biology and function.
A comprehensive spatial immune atlas identifies 11 immune populations and shows increased macrophages, plasma cells, and especially B cells in DKD; B cells form organized, DKD-associated microenvironments enriched in CXCL13–CXCR5 signaling, suggesting TLS-like kidney structures.
DKD shifts niche composition by reducing PT and DCT niches while expanding immune and injured tubular niches; niche frequencies correlate with eGFR, implying potential functional indicators of kidney health and disease severity.
A Nature paper briefing highlights single-cell gene-expression profiling and spatial mapping that identify immune microenvironments, notably B cell–rich regions, in a subset of individuals with accelerated DKD progression.
The briefing places findings in the broader nephrology context, referencing prior work in Nature Medicine, JCI, and Nature Genetics, and notes access/licensing details for Nature content while conveying core discovery and implications.
B cell–dominant microenvironments cluster within profibrotic tubular niches and align with injury and fibrosis; imaging mass cytometry shows abundant CD20+ B and CD4+ T cells, with naive and memory B cells and in situ activation markers.
Summary based on 2 sources
Get a daily email with more Science stories
Sources
Nature • Apr 29, 2026
Spatial atlas of diabetic kidney disease reveals a B cell-rich subgroup
Nature • Apr 29, 2026
A cell atlas charts the immune architecture of diabetic kidney disease