New Atlas Maps Maternal-Fetal Interface, Unveils Placental Dynamics and Complications
April 8, 2026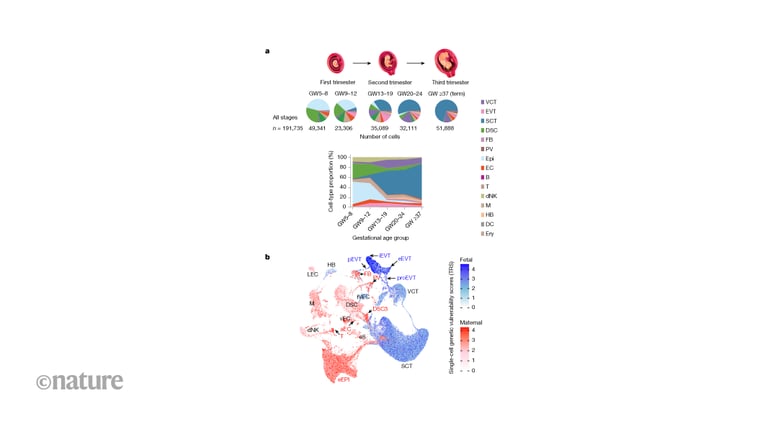
A comprehensive, spatially resolved atlas maps the human maternal–fetal interface across gestation, integrating multiomics and spatial transcriptomics to illuminate regulatory networks, cellular dynamics, and placental architecture.
Using high-resolution single-nucleus multiomics and spatial mapping, the study identifies cell types, regulatory networks, spatial niches, and dynamic processes driving placental development and its complications.
A bistable toggle-switch model of trophoblast fate reveals a mutual repression between EVT and SCT lineages, with lineage-specific transcription factors steering commitment.
Tracked EVT aggregation around uterine spiral arteries shows endothelial state transitions from canonical arterial endothelial cells through primed and identity-loss stages to apoptosis, culminating in EVT-driven vessel remodeling and endothelium displacement.
Major cell classes in decidua basalis and basal plate are annotated with maternal or fetal origin for over 95% of cells, highlighting molecular divergence between lineages in key cell types.
Newly discovered regulatory cells may act as a “speed bump” to modulate placental invasion, potentially linking to conditions such as preeclampsia and placenta accreta.
Deep-coverage snRNA-seq reveals intermediate trophoblast states and, via pseudotime, bifurcation of VCTs into SCTs and EVTs with distinct intermediate states emerging across gestation.
Researchers describe this work as a foundation, emphasizing the need to study healthy versus complicated pregnancies with more cells and samples for broader coverage.
References draw on decades of obstetrics and placental biology research, underscoring a rich lineage of prior single-cell and pregnancy studies.
The findings are based on Wang et al.’s Nature article, with licensing for Figure 1 under CC BY 4.0.
A 0.5-micron-resolution Stereo-seq atlas maps 1.1 million cells across 16 basal plate sections, revealing six spatial communities that map to decidual, vascular, maternal–fetal junction, and floating villus regions.
The map highlights cell states vulnerable in common pregnancy complications, offering mechanistic insight and potential targets for monitoring or intervention.
Summary based on 3 sources
Get a daily email with more Science stories
Sources

Nature • Apr 8, 2026
Single-cell spatiotemporal dissection of the human maternal–fetal interface
Nature • Apr 8, 2026
Mapping the maternal–fetal interface through pregnancy in high resolution
Live Science • Apr 8, 2026
'No one knows what they are': Researchers discover new type of cell that's seen only during pregnancy