93-Gene Signature Unravels Unique Pathways in Pediatric Brain Tumors: New Study Reveals Oncogenic Mechanisms
March 25, 2026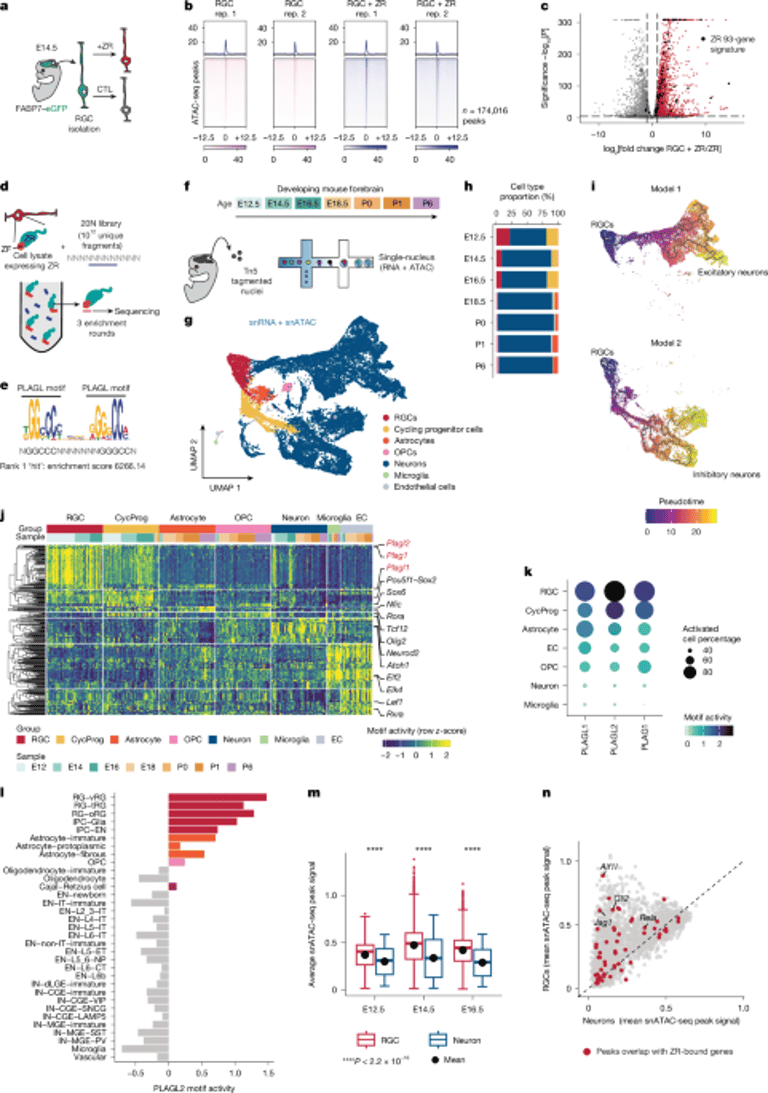
A 93-gene ZR signature can stratify ZR-driven tumors in human PF-EPNs and ZR EPNs, while PF-EPNs and PLAG/L fusions show distinct gene expression signatures and motif enrichments.
ZR and PLAG/L fusions in human and mouse models converge on PLAG/L motif–driven chromatin programs, yet ZR-specific genes such as L1CAM, CCND1, and WNT3A reveal signatures unique to ZR EPNs, indicating convergent but distinct oncogenic paths.
Ongoing Nature coverage highlights continued research into embryonic brain development and pediatric brain tumors.
The study is framed around Kardian, A. S. et al., Dominant clones leverage developmental epigenomic states to drive ependymoma, published in Nature in 2026.
Analyses focus on accessible chromatin sites to understand how ZFTA–RELA interacts with developmental epigenomic states.
ZFTA–RELA, a cancer-promoting fusion, binds chromatin modules accessible during embryonic brain development in developing mice, pointing to at-risk cell lineages for transformation.
Conclusion: paediatric fusion oncoproteins exploit specific developmental epigenomic modules to initiate and sustain tumor heterogeneity, with dominant progenitor-like clones driving tumor formation.
ZR activity persists beyond normal windows, keeping PLAG/L motif accessibility and proliferation signals in cycling progenitors, while differentiated-like ZR EPN cells are less proliferative.
The article notes access options for the full Nature piece, including subscription and licensing details.
ZR-driven EPNs exhibit diverse cell states (RGC-like, cycling progenitor-like, neuronal-like) with a differentiation block, while YM-driven EPNs resemble RGC-like states and OPC involvement is limited in ZR EPN.
PLAG/L motifs are more accessible in cycling progenitors and are hijacked by ZR and PLAG/L fusions to rewire the epigenome, with a binding preference for PLAG/L motifs and a core GGGCC sequence.
Barcoded tracking shows early clonal diversity in ZR tumors, followed by a dominant clone that recapitulates multiple developmental lineages, suggesting dominant progenitor-like cells establish tumor heterogeneity.
Summary based on 2 sources
Get a daily email with more Science stories
Sources
Nature • Mar 25, 2026
Dominant clones leverage developmental epigenomic states to drive ependymoma
Nature • Mar 25, 2026
A cancer-promoting fusion protein acts during embryonic brain development